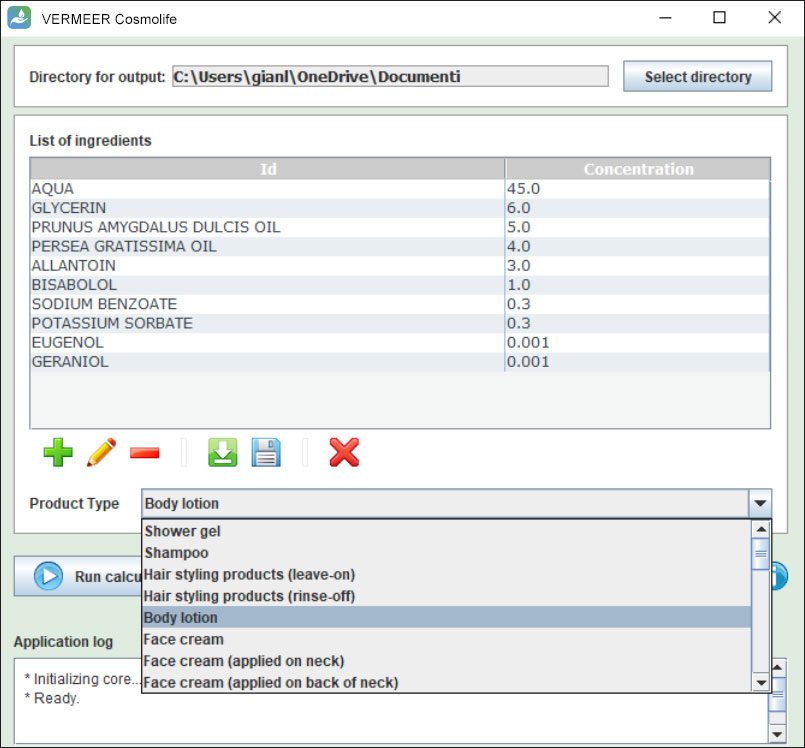
Input data
VERMEER Cosmolife has a user-friendly graphical interface, in which the user adds the structures of interest and runs the calculations. The user can insert single or multiple structures. These structures can be entered by SMILES, CAS or INCI. The software searches the structure in a database implemented within the software.
Another basic input information to be included is the concentration of the ingredient.
Lastly, in a drop-down menu, the user has to insert the specific product type to define an exposure scenario. Moreover, the user has the possibility to decide the directory for output.
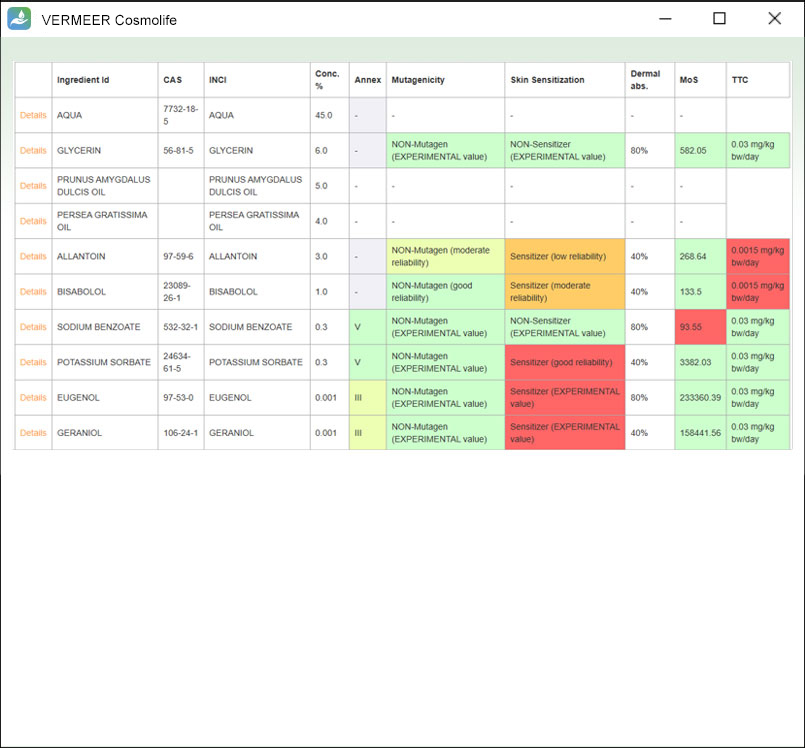
Summary output table
The software provides a preliminary output table with a summary of the hazard and exposure features of the ingredients, previously selected in the input file.
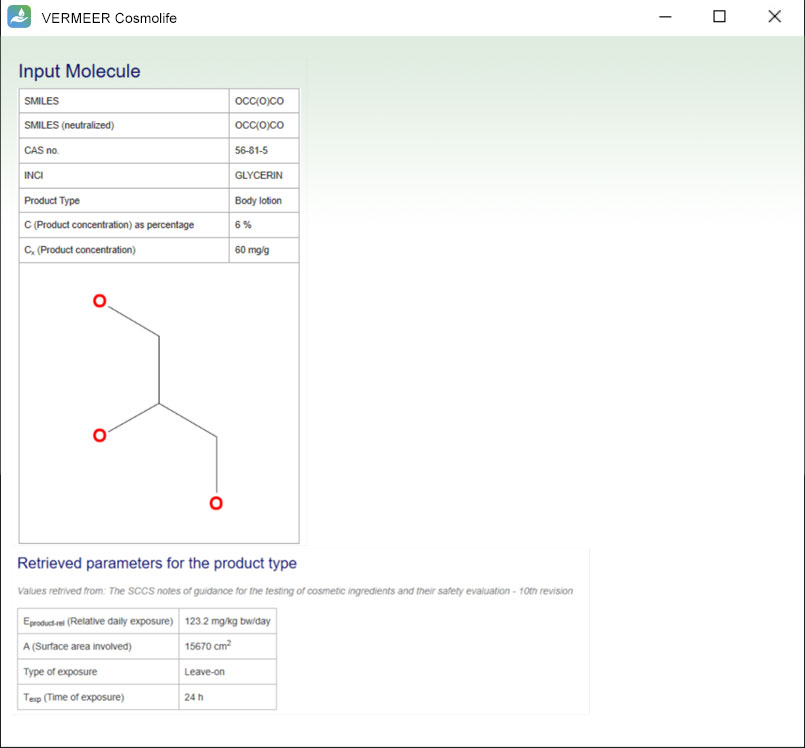
Information about input molecule and retrieved parameters for the product type
A summary of the input parameters is given. Moreover, the software shows the parameters related to the selected product type, retrieved from the SCCS Notes of Guidance.
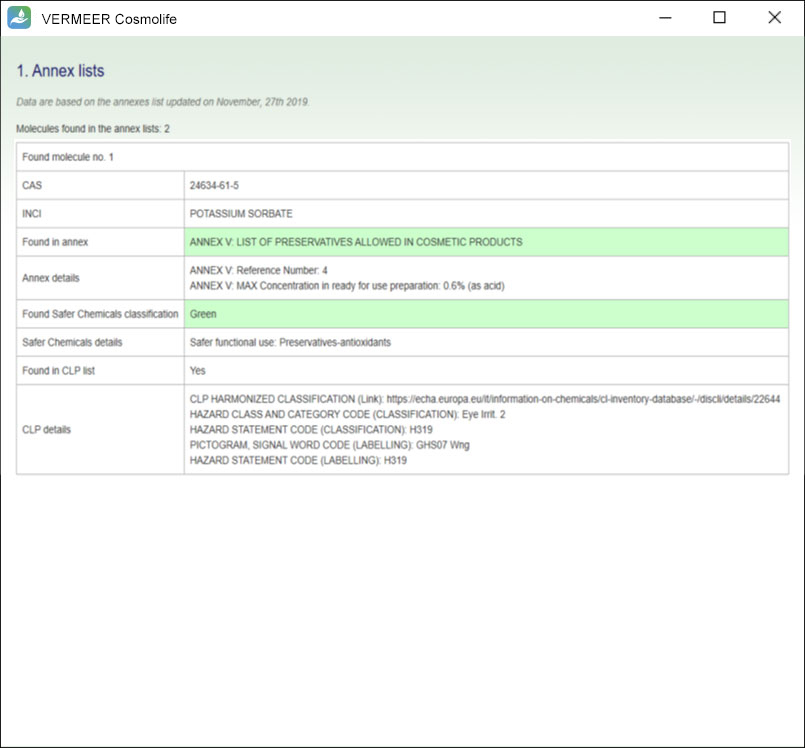
Annex lists
The software provides the check with the Annexes of the Regulation (EC) n°1223/2009. The database implemented within the software contains these lists, with all the information reported in the Regulation, which are the maximum concentration, the product type in which the ingredient is allowed or the wording of conditions of use and warning.
Moreover, in this section the software provides a check with the Classification, Labelling and Packaging (CLP) Regulation and a check with the Safer Chemical Ingredients List (SCIL).
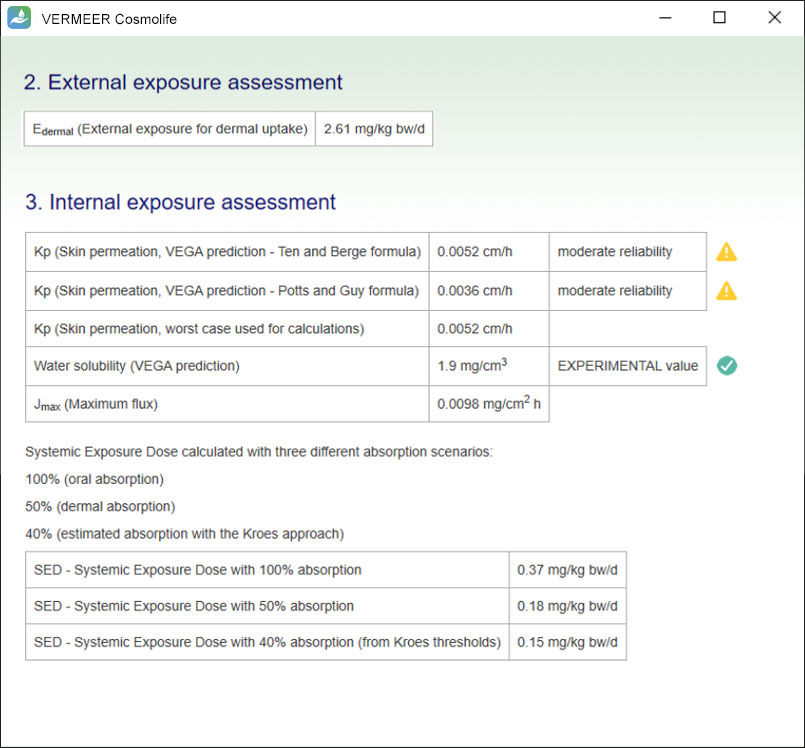
Exposure assessment
VERMEER Cosmolife calculates the external dermal exposure using the formulas proposed within the SCCS Notes of Guidance.
Moreover, the systemic exposure dose (SED) is calculated, using the formulas proposed within the SCCS Notes of Guidance and a new approach developed using specific models for skin permeation.
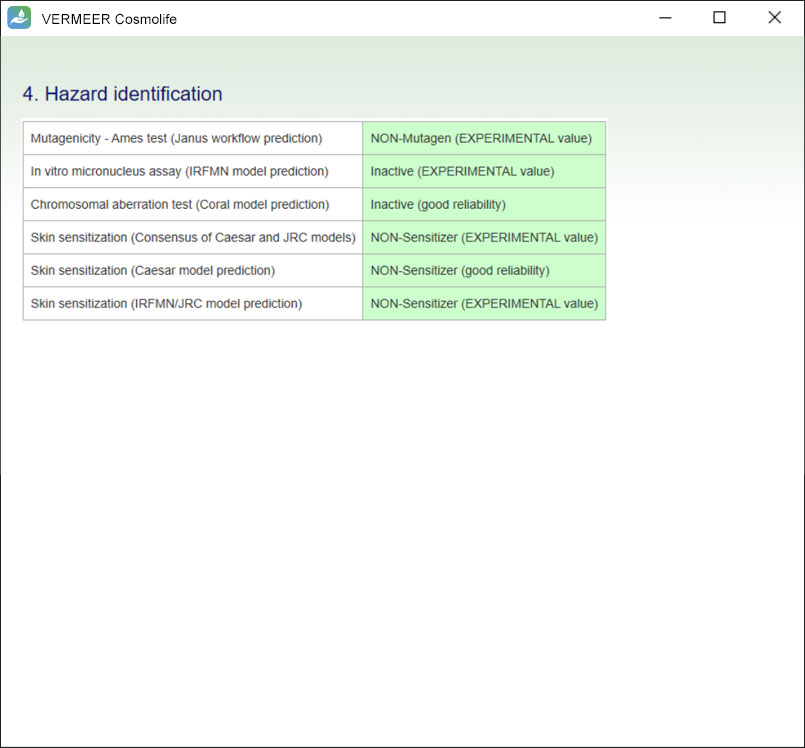
Hazard identification
The software reports values (experimental if available or predicted) related to some toxicological endpoints.
At the moment the software provides an evaluation of mutagenicity/genotoxicity and skin sensitization.
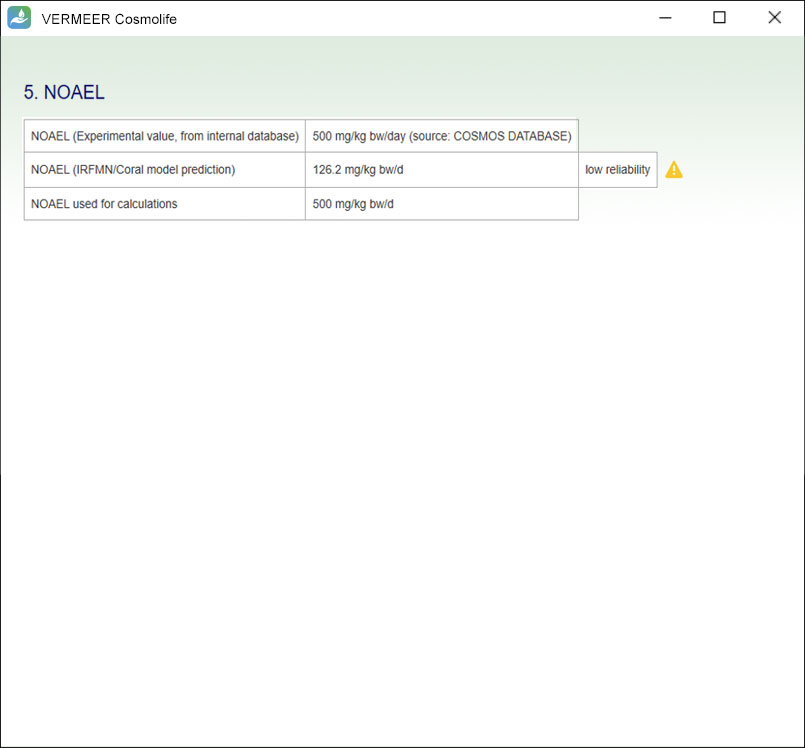
NOAEL
VERMEER Cosmolife provides the fundamental value of the No Observed Adverse Effect Level (NOAEL). The software searches in the internal database experimental values for NOAEL or predict this value using the CORAL NOAEL model implemented in VEGA. If present, the experimental value will be used for risk characterization.
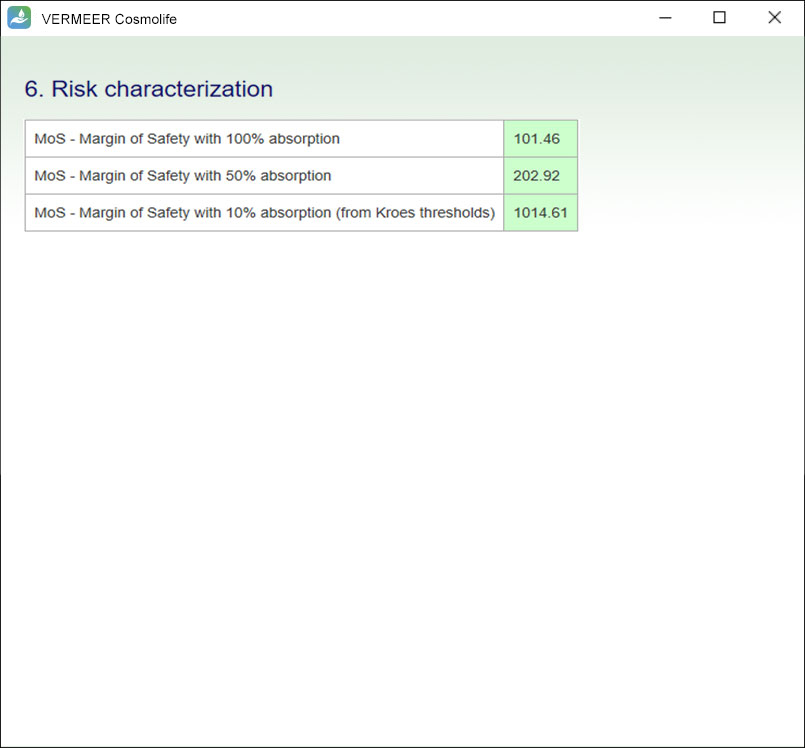
Risk characterization
The software carries out a risk characterization, considering the process proposed within the SCCS Notes of Guidance. For cosmetics, the focus is on systemic toxicity and the Margin of Safety (MoS) is calculated.
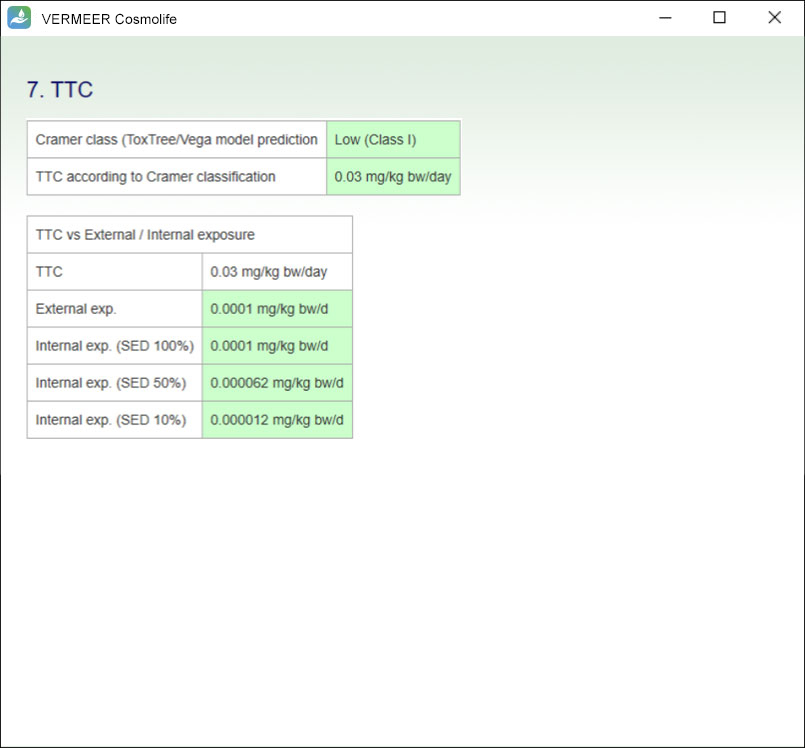
TTC assessment
The software includes the assessment considering the TTC approach. Moreover, the software compares the external and internal exposure values, previously obtained, with the TTC threshold related to the specific ingredient, in order to incorporate dermal absorption into the use of TTC.
VERMEER Cosmolife Screenshots
